Metabolism Can Be Seen as the Process of Converting
Energy is trapped in the chemic bonds of nutrient molecules. How is it then made usable for cellular functions and biosynthetic processes?
Where does the free energy that makes life possible come from? Humans obtain energy from three classes of fuel molecules: carbohydrates, lipids, and proteins. The potential chemical free energy of these molecules is transformed into other forms, such equally thermal, kinetic, and other chemical forms.
Nutrients of Human being Metabolism
Carbohydrates, lipids, and proteins are the major constituents of foods and serve as fuel molecules for the homo body. The digestion (breaking down into smaller pieces) of these nutrients in the comestible tract and the subsequent absorption (entry into the bloodstream) of the digestive finish products make it possible for tissues and cells to transform the potential chemical free energy of food into useful work.
The major captivated finish products of nutrient digestion are monosaccharides, mainly glucose (from carbohydrates); monoacylglycerol and long-chain fatty acids (from lipids); and modest peptides and amino acids (from protein). One time in the bloodstream, different cells can metabolize these nutrients. We accept long known that these three classes of molecules are fuel sources for human metabolism, all the same it is a mutual misconception (especially among undergraduates) that man cells utilize merely glucose as a source of energy. This misinformation may arise from the manner most textbooks explain energy metabolism, emphasizing glycolysis (the metabolic pathway for glucose deposition) and omitting fatty acid or amino acid oxidation. Here we discuss how the 3 nutrients (carbohydrates, proteins, and lipids) are metabolized in man cells in a style that may help avoid this oversimplified view of the metabolism.
Historical Overview of Free energy Metabolism

During the eighteenth century, the initial studies, adult by Joseph Black, Joseph Priestley, Carl Wilhelm Scheele, and Antoine Lavoisier, played a special office in identifying two gases, oxygen and carbon dioxide, that are cardinal to energy metabolism. Lavoisier, the French nobleman who owns the title of "father of modern chemistry," characterized the composition of the air we breathe and conducted the first experiments on energy conservation and transformation in the organism.
I of Lavoisier'due south master questions at this fourth dimension was: How does oxygen's function in combustion relate to the process of respiration in living organisms? Using a calorimeter to make quantitative measurements with republic of guinea pigs and later on with himself and his banana, he demonstrated that respiration is a slow class of combustion (Figure 1). Based on the concept that oxygen burned the carbon in nutrient, Lavoisier showed that the exhaled air contained carbon dioxide, which was formed from the reaction between oxygen (present in the air) and organic molecules inside the organism. Lavoisier too observed that oestrus is continually produced by the trunk during respiration. It was and so, in the eye of the nineteenth century, that Justus Liebig conducted animal studies and recognized that proteins, carbohydrates, and fats were oxidized in the body. Finally, pioneering contributions to metabolism and nutrition came from the studies of a Liebig's protégé, Carl von Voit, and his talented pupil, Max Rubner. Voit demonstrated that oxygen consumption is the result of cellular metabolism, while Rubner measured the major energy value of certain foods in lodge to calculate the caloric values that are still used today. For example, carbohydrates and proteins produce approximately 4 kcal/grand of energy, whereas lipids can generate up to 9 kcal/g. Rubner'southward observations proved that, for a resting animal, heat product was equivalent to heat elimination, confirming that the law of conservation of free energy, implied in Lavoisier's early experiments, was applicable to living organisms likewise. Therefore, what makes life possible is the transformation of the potential chemical energy of fuel molecules through a serial of reactions within a cell, enabled past oxygen, into other forms of chemical energy, motion energy, kinetic energy, and thermal free energy.
Energy Conservation: Mechanisms of ATP Synthesis
Free energy metabolism is the general procedure past which living cells learn and use the energy needed to stay alive, to grow, and to reproduce. How is the energy released while breaking the chemical bonds of food molecules captured for other uses by the cells? The answer lies in the coupling between the oxidation of nutrients and the synthesis of high-energy compounds, particularly ATP, which works as the master chemical free energy carrier in all cells.
There are two mechanisms of ATP synthesis: 1. oxidative phosphorylation, the procedure by which ATP is synthesized from ADP and inorganic phosphate (Pi) that takes identify in mitochondrion; and ii. substrate-level phosphorylation, in which ATP is synthesized through the transfer of high-energy phosphoryl groups from loftier-free energy compounds to ADP. The latter occurs in both the mitochondrion, during the tricarboxylic acid (TCA) cycle, and in the cytoplasm, during glycolysis. In the next section, we focus on oxidative phosphorylation, the primary mechanism of ATP synthesis in about of homo cells. Afterward we comment on the metabolic pathways in which the iii classes of nutrient molecules are degraded
Oxidative Phosphorylation: The Main Mechanism of ATP Synthesis in Most Human Cells
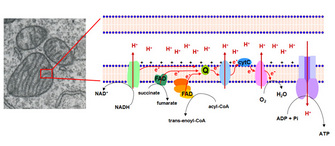
The metabolic reactions are energy-transducing processes in which the oxidation-reduction reactions are vital for ATP synthesis. In these reactions, the electrons removed by the oxidation of fuel molecules are transferred to ii major electron carrier coenzymes, nicotinamide adenine dinucleotide (NAD+) and flavin adenine dinucleotide (FAD), that are converted to their reduced forms, NADH and FADH2. Oxidative phosphorylation depends on the electron send from NADH or FADHtwo to O2, forming H2O. The electrons are "transported" through a number of protein complexes located in the inner mitochondrial membrane, which contains fastened chemical groups (flavins, iron-sulfur groups, heme, and cooper ions) capable of accepting or donating one or more electrons (Figure 2). These protein complexes, known as the electron transfer system (ETS), allow distribution of the free energy betwixt the reduced coenzymes and the O2 and more efficient free energy conservation.
The electrons are transferred from NADH to O2 through three protein complexes: NADH dehydrogenase, cytochrome reductase, and cytochrome oxidase. Electron transport between the complexes occurs through other mobile electron carriers, ubiquinone and cytochrome c. FAD is linked to the enzyme succinate dehydrogenase of the TCA bike and another enzyme, acyl-CoA dehydrogenase of the fatty acid oxidation pathway. During the reactions catalyzed by these enzymes, FAD is reduced to FADHtwo, whose electrons are and then transferred to O2 through cytochrome reductase and cytochrome oxidase, as described for NADH dehydrogenase electrons (Figure 2).
The electron transfer through the components of ETS is associated with proton (H+) pumping from the mitochondrial matrix to intermembrane infinite of the mitochondria. These observations led Peter Mitchell, in 1961, to propose his revolutionary chemiosmotic hypothesis. In this hypothesis, Mitchell proposed that H+ pumping generates what he called the proton motive forcefulness, a combination of the pH gradient across the inner mitochondrial membrane and the transmembrane electrical potential, which drives the ATP synthesis from ADP and Pi. ATP is synthesized past the ATP synthase complex, through which H+ protons render to the mitchondrial matrix (Effigy 2, far right). Paul Boyer starting time described the ATP synthase catalytic machinery and showed both that the energy input from the H+ slope was used for ATP release from the catalytic site, and that the three active sites of the enzyme worked cooperatively in such a way that ATP from 1 site could not exist released unless ADP and Pi were available to bind to some other site.
Oxidation of Carbohydrates, Proteins, and Fats Converge on the Tricarboxylic Acid Wheel

Interconversion of free energy between reduced coenzymes and Otwo directs ATP synthesis, but how (and where) are NADH and FADH2 reduced? In aerobic respiration or aerobiosis, all products of nutrients' degradation converge to a key pathway in the metabolism, the TCA cycle. In this pathway, the acetyl grouping of acetyl-CoA resulting from the catabolism of glucose, fatty acids, and some amino acids is completely oxidized to CO2 with concomitant reduction of electron transporting coenzymes (NADH and FADH2). Consisting of eight reactions, the bicycle starts with condensing acetyl-CoA and oxaloacetate to generate citrate (Figure 3). The next seven reactions regenerate oxaloacetate and include four oxidation reactions in which energy is conserved with the reduction of NAD+ and FAD coenzymes to NADH and FADHtwo, whose electrons will then be transferred to O2 through the ETS. In improver, a GTP or an ATP molecule is direct formed as an example of substrate-level phosphorylation. In this instance, the hydrolysis of the thioester bail of succinyl-CoA with concomitant enzyme phosphorylation is coupled to the transfer of an enzyme-bound phosphate group to GDP or ADP. Importantly, although O2 does not participate directly in this pathway, the TCA cycle only operates in aerobic conditions because the oxidized NAD+ and FAD are regenerated only in the ETS. Likewise noteworthy is that TCA cycle intermediates may also exist used as the precursors of different biosynthetic processes.
The TCA wheel is likewise known as the Krebs bicycle, named afterward its discoverer, Sir Hans Kreb. Krebs based his conception of this wheel on four main observations made in the 1930s. The commencement was the discovery in 1935 of the sequence of reactions from succinate to fumarate to malate to oxaloacetate past Albert Szent-Gyorgyi, who showed that these dicarboxylic acids present in animal tissues stimulate O2 consumption. The second was the finding of the sequence from citrate to α-ketoglutarate to succinate, in 1937, by Carl Martius and Franz Knoop. Side by side was the observation by Krebs himself, working on muscle piece cultures, that the addition of tricarboxylic acids even in very low concentrations promoted the oxidation of a much higher amount of pyruvate, suggesting a catalytic event of these compounds. And the fourth was Krebs'south observation that malonate, an inhibitor of succinate dehydrogenase, completely stopped the oxidation of pyruvate by the add-on of tricarboxylic acids and that the add-on of oxaloacetate in the medium in this condition generated citrate, which accumulated, thus elegantly showing the cyclic nature of the pathway.
Pathways for Nutrient Degradation that Converge onto the TCA Wheel
Glycolysis

Glycolysis is the pathway in which one glucose molecule is degraded into ii pyruvate molecules. Interestingly, during the initial phase, energy is consumed considering two ATP molecules are used upward to activate glucose and fructose-6-phosphate. Part of the energy derived from the breakdown of the phosphoanhydride bond of ATP is conserved in the formation of phosphate-ester bonds in glucose-6-phosphate and fructose-one,6-biphosphate (Effigy 4).
In the second role of glycolysis, the bulk of the complimentary energy obtained from the oxidation of the aldehyde group of glyceraldehyde 3-phosphate (G3P) is conserved in the acyl-phosphate grouping of 1,3- bisphosphoglycerate (1,3-BPG), which contains loftier gratis energy. Then, part of the potential free energy of i,3BPG, released during its conversion to three-phosphoglycerate, is coupled to the phosphorylation of ADP to ATP. The second reaction where ATP synthesis occurs is the conversion of phosphoenolpyruvate (PEP) to pyruvate. PEP is a high-energy compound due to its phosphate-ester bail, and therefore the conversion reaction of PEP to pyruvate is coupled with ADP phosphorylation. This mechanism of ATP synthesis is called substrate-level phosphorylation.
For complete oxidation, pyruvate molecules generated in glycolysis are transported to the mitochondrial matrix to be converted into acetyl-CoA in a reaction catalyzed past the multienzyme complex pyruvate dehydrogenase (Figure v). When Krebs proposed the TCA bike in 1937, he thought that citrate was synthesized from oxaloacetate and pyruvate (or a derivative of it). Just after Lipmann's discovery of coenzyme A in 1945 and the subsequent work of R. Stern, S. Ochoa, and F. Lynen did it become clear that the molecule acetyl-CoA donated its acetyl group to oxaloacetate. Until this time, the TCA cycle was seen as a pathway to sugar oxidation only. Well-nigh high school textbooks reflect this flow of biochemistry knowledge and do not emphasize how the lipid and amino acid degradation pathways converge on the TCA cycle.
The Fat Acid Oxidation Pathway Intersects the TCA Cycle
In 1904, Knoop, in a archetype experiment, decisively showed that fatty acid oxidation was a process by which two-carbon units were progressively removed from the carboxyl end fatty acid molecule. The process consists of four reactions and generates acetyl-CoA and the acyl-CoA molecule shortened by two carbons, with the concomitant reduction of FAD by enzyme acyl-CoA dehydrogenase and of NAD+ by β-hydroxyacyl-CoA dehydrogenase. This pathway is known as β-oxidation because the β-carbon atom is oxidized prior to when the bond betwixt carbons β and α is cleaved (Effigy vi). The iv steps of β-oxidation are continuously repeated until the acyl-CoA is entirely oxidized to acetyl-CoA, which then enters the TCA bike. In the 1950s, a series of experiments verified that the carbon atoms of fatty acids were the same ones that appeared in the acids of TCA cycle.
Amino Acrid Transamination/Deamination Contributes to the TCA Cycle

2 points must exist considered regarding the utilize of amino acids as fuels in energy metabolism. The first is the presence of nitrogen in amino acid composition, which must be removed before amino acids go metabolically useful. The other is that in that location are at to the lowest degree twenty different amino acids, each of which requires a different degradation pathway. For our purpose here, it is of import to mention two kinds of reactions involving amino acrid: transamination and deamination. In the outset kind of reaction, the enzymes aminotransferases catechumen amino acids to their respective α-ketoacids by transferring the amino group of one amino acrid to an α-ketoacid. This reaction allows the amino acids to be interconverted. The 2nd kind of reaction, deamination, removes the amino group of the amino acrid in the form of ammonia. In the liver, the oxidative deamination of glutamate results in α-keto-glutarate (a TCA cycle intermediate) and ammonia, which is converted into urea and excreted. Deamination reactions in other organs form ammonia that is generally incorporated into glutamate to generate glutamine, which is the main transporter of amino groups in blood. Hence, all amino acids through transamination/deamination reactions can be converted into intermediates of TCA cycle, directly or via conversion to pyruvate or acetyl-CoA (Figure 5).
Summary
The transformation of the chemical energy of fuel molecules into useful energy is strictly regulated, and several factors control the utilise of glucose, fat acids, and amino acids by the different cells. For example, not all cells take the enzyme machinery and the proper cellular compartments to utilize all three fuel molecules. Red blood cells are devoid of mitochondria and are therefore unable to oxidize neither fat acids nor amino acids, relying simply on glucose for ATP synthesis. In addition, even in cells that tin use all nutrients, the type of nutrient substrate that is oxidized changes according to the physiological situation of the cell, such as the fed and fasting states. Different signals dictate how cells can adapt to each situation, such as hormones, which may exert powerful effects by switching key enzyme activities in a matter of seconds, or how they may modulate gene expression profile, irresolute the whole cell metabolic contour. We must therefore understand all metabolic pathways every bit integrated events controlling energy regulation and conversion
References and Recommended Reading
Blaxter, K. Free energy Metabolism in Animals and Human. Cambridge: Cambridge Academy Printing, 1989.
Holmes, F. L. Lavoisier and the Chemical science of Life. Madison: Academy of Wisconsin Press, 1985.
Krebs, H. Nobel Prize Lecture (1953). Nobelprize.org, 2010.
Kresge, Due north., Simoni, R. D., & Hill, R. L. ATP synthesis and the bounden change mechanism: The piece of work of Paul D. Boyer. Journal of Biological Chemistry 281, e18 (2006).
Lusk, G. The Elements of the Science of Nutrition, quaternary ed. Philadelphia: West. B. Saunders, 1931.
Luz, M. R. M. P. Glucose as the sole metabolic fuel: A study on the possible influence of teachers' knowledge on the establishment of a misconception among Brazilian high schoolhouse stucents. Advances in Physiological Instruction 32, 225–230 (2008) doi:10.1152/advan.00050.2007.
Luz, M. R. M. P. et al. Glucose as the sole metabolic fuel: The possible influence of formal teaching on the establishment of a misconception nigh the energy-yielding metabolism among Brazilian students. Biochemistry and Molecular Biological science Education 36, 407–416 (2008) doi:10.1002/bmb.20235.
Oliveira, G. A. et al. Students' misconception nigh free energy yielding metabolism: Glucose as the sole metabolic fuel. Advances in Physiological Education 27, 97–101 (2003 doi:10.1152/advan.00009.2003.
Source: https://www.nature.com/scitable/topicpage/nutrient-utilization-in-humans-metabolism-pathways-14234029/
0 Response to "Metabolism Can Be Seen as the Process of Converting"
Post a Comment